Hyperhidrosis - an unknown widespread "silent" disorder
Alma Rystedt1, Kerstin Brismar2, Sten-Magnus Aquilonius3, Hans Naver4, Carl Swartling1,5*
1Hidrosis Clinic, Stockholm, Sweden
2Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm, Sweden
3Department of Neuroscience, Neurology, Uppsala University, Uppsala University Hospital, Uppsala, Sweden
4Department of Internal Medicine, Neurology, Nyköping Hospital, Nyköping, Sweden
5Department of Medical Sciences, Dermatology and Venereology, Uppsala University, Uppsala, Sweden
Abstract
Hyperhidrosis is a social, emotional and occupational disability which affects close to 3 % of the population. Patients with hyperhidrosis suffer an extremely negative impact on their quality of life on a par with being severely affected by psoriasis. Most of the sufferers have the primary genetic form of hyperhidrosis. Secondary hyperhidrosis can often be omitted based on anamnestic data, but sometimes further examinations must be performed.
Topical treatment (e.g. aluminium chloride) is the first choice for localised hyperhidrosis. Botulinum toxin, iontophoresis, microwave thermolysis (miraDry®), and/or systemic medications are indicated if topical treatment is insufficient or not applicable. Endoscopic Thoracic Sympathectomy (ETS) is no longer performed in Sweden due to the serious side effects profile. In countries where ETS still is performed, patients must be carefully selected and educated to fully understand the possibility of limited efficacy and the risks of complications including, but not limited to, compensatory sweating. This treatment should be the last option.
Examination- and treatment recommendations based on international guidelines and literature are presented in this review.
Hyperhidrosis – symptom or disorder?
The question is important since it highlights the view that the patient, surrounding people and the care provider have of the condition. Along with other physiologically-inadequate and troubling somatic reactions such as tachycardia, palpitations or disturbances to the gastrointestinal tract, the severity of the symptom can vary but if an individual has disabling symptoms, the condition is called a disorder of which IBS (irritable bowel syndrome) is one example. In this review, we highlight hyperhidrosis as the “silent” disorder, a widespread disorder which is not discussed on courses for doctors and care providers and about which there is a low level of knowledge within the profession as a consequence. This overview article will highlight the diagnosis, the patients and the treatment options.
The function and pathology of sweating
Sweating is the most important effector in thermoregulation and is controlled through the hypothalamus1. Sweat on the palms of the hands and soles of the feet help to provide a good grip, which has been important to human beings during evolution and having normal palm moisture is important to us when doing widely differing activities such as handicrafts, handling paper and sport. The palmoplantar sweating, sometimes called “emotional”, is controlled through the cortex, the limbic system and through sympathetic (fight and flight) nerves (Figure 1)1
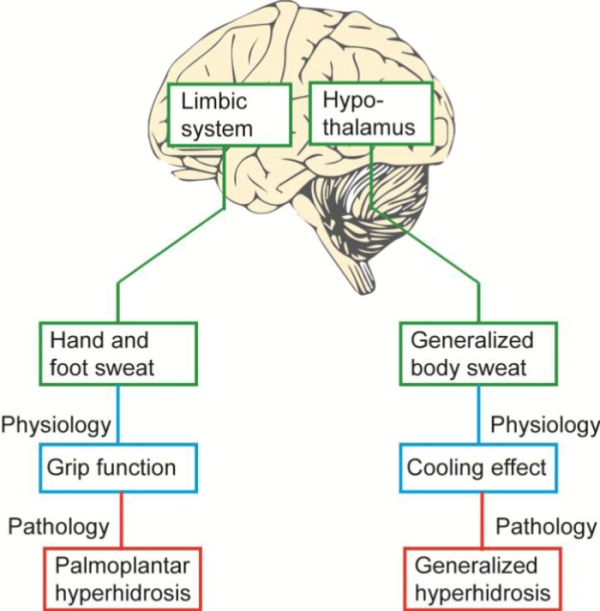
Figure 1: We sweat partly to cool our body and partly to obtain a good grip function. These functions are controlled from different parts of the “old” part of the brain: grip function from the cortex and the limbic system, thermoregulation from nuclei in the hypothalamus. Since the hypothalamus is also a nucleus of the limbic system, factors such as stress also usually draw sweat from the whole of the body, and heat/exertion can make hand and foot sweat worse. What may be physiological can transform into pathological in 2.8 % who are genetically predisposed. The figure is reprinted with permission from Läkartidningen2.
Reflexes that are triggered by pressure on the palm of the hand/sole of the foot can trigger palmoplantar sweating.
The eccrine sweat glands with cholinergic muscarinic receptors receive signals from sympathetic fibres with acetylcholine as the signal substance. The co-transmitters CGRP and VIP are potent vasodilators and lead to greater vascular permeability which is important when sweat is being produced. After NaCl has been reabsorbed into the sweat duct, the eccrine sweat turns into a hypoosmolar salt solution. The presence of cystic fibrosis means that this reabsoption capacity is lacking, leading to extra salty sweat, which is fairly often observed by parents of children with the disease.
Armpits and groins have three types of sweat glands: eccrine, apocrine and hybrid apoeccrine (Figure 2).
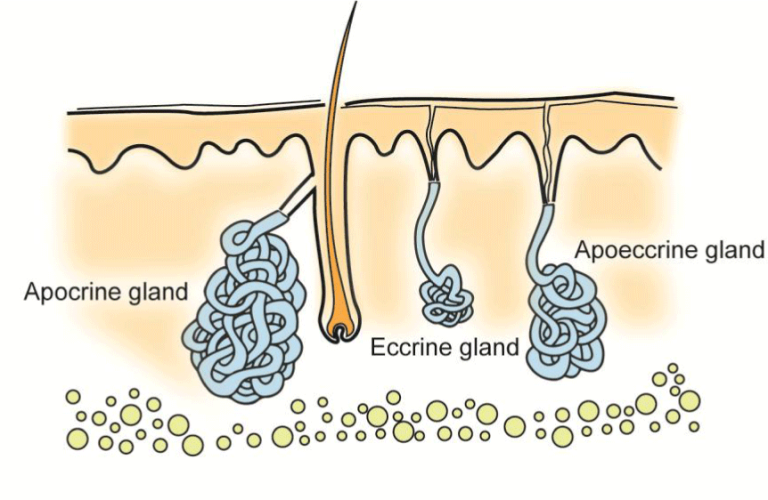
Figure 2: The armpits and groins have 3 different types of sweat glands which distinguish themselves histologically and functionally. The eccrine glands make the ample sweat which consists of a salt solution. The apocrine sweat constitutes a small amount of oily liquid which contains pheromones and, when it breaks down skin bacteria, gives rise to the typical odour of sweat. The apoeccrine gland is a hybrid which is similar in function to the eccrine and which can produce large quantities of sweat in the form of a salt solution. The figure is reprinted with permission from Läkartidningen2.
The eccrine and apoeccrine sweat glands produce the salt solution, “the normal sweat”. The apocrine glands have some features of mammary gland secretion as the sweat is energy-rich. The sweat consists of a small quantity which is of an oily consistency. The apocrine sweat is primarily odour-free but the characteristic smell of sweat occurs when it breaks down skin bacteria in armpits and groins. A pronounced smell of sweat with its impact on an individual’s quality of life is called bromhidrosis. The apocrine sweat contains pheromones whose odour signals may also be significant as regards sexual attraction in humans3.
Hyperhidrosis is characterised by an abnormal response to heat, exertion and stress with pronounced sweating either in general or focally. This can be seen as an extension of the physiological response where general hyperhidrosis involves “the thermostat”, the hypothalamus, and focal, symmetrical hyperhidrosis cortex and the limbic system. Patients with hyperhidrosis display increased activity in the sympathetic nervous system (sudomotor) on arousal (pain, shouting, caressing)4.
Primary hyperhidrosis
A major American study shows that 2.8 % of the population suffers from hyperhidrosis5. The majority have the primary form which is hereditary, probably autosomally inherited with incomplete penetrance6. It can be divided into focal and general primary hyperhidrosis. The focal is bilaterally symmetrical: hands, feet, axillae or groins. Focal hyperhidrosis from the face/head does occur but is often part of the general form. Generalized sweating usually involves both the head and trunk and in severe cases also extremities and groins/glutes. Combined focal and general hyperhidrosis does occur. Other common combinations of focal hyperhidrosis are hands and feet, hands, feet and axillae, and groins and axillae. Only 25 % of the patients at the Hidrosis Clinic, Stockholm, Sweden, suffer from hyperhidrosis from one area and 50 % from two or three areas, while 25% sweat abnormally from four or more areas (unpublished data from 2010). These numbers are on parity with statistics from the hidrosis clinic at Uppsala University Hospital, Uppsala, Sweden.
Hyperhidrosis from the hands and feet usually starts during early childhood but axillary hyperhidrosis often starts in the teenage years. Many people who have general hyperhidrosis start after the age of 50. For many women, if it starts late in life it is called postmenopausal hyperhidrosis, even if other climacteric symptoms are missing and oestrogen substitution is ineffectual7.
Patients with general hyperhidrosis say that heat/exertion is the most deteriorative factor, and stress is the second most deteriorative. The reverse applies to focal hyperhidrosis from hands and feet where stress is the most deteriorative and heat/exertion come second. The fact that the “thermostat”, the hypothalamus, is part of the limbic system may explain why the deteriorative factors of heat/exertion and stress covary with general and focal hyperhidrosis. The prognosis of hyperhidrosis is not known. For some, the problems pass but for many they remain for life. The disorder can also change in character; for example, it can start as axillary hyperhidrosis in the teenage years, followed by a problem-free interval and later general hyperhidrosis in the 60s. See table 1 for the diagnosis of primary focal hyperhidrosis.
| Suggests secondary hyperhidrosis | |
| short anamnesis | |
| symptoms of another disease which can give rise to secondary hyperhidrosis | |
| regional or asymmetrical sweating | |
| Suggests primary hyperhidrosis | |
| long anamnesis | |
| early onset | |
| heredity | |
| focal, bilateral symmetrical sweating | |
| sweating stops at night | |
Table 1: In most cases, it is easy to exclude or diagnose secondary hyperhidrosis without taking samples and performing examinations. The table is reprinted with permission from Läkartidningen2.
Hyperhidrosis has an extremely negative impact on quality of life. The Dermatology Life Quality Index (DLQI) can be used to objectivise this, to assess treatment results and compare results with other skin diseases which are surveyed using the same questionnaire. We see that patients with hyperhidrosis can have a heavily reduced quality of life on a par with the most severe psoriasis patients who are surveyed using the same questionnaire (Figure 3). At the specialist hidrosis clinics in Stockholm and Uppsala the DLQI results are equal to those of these published studies.
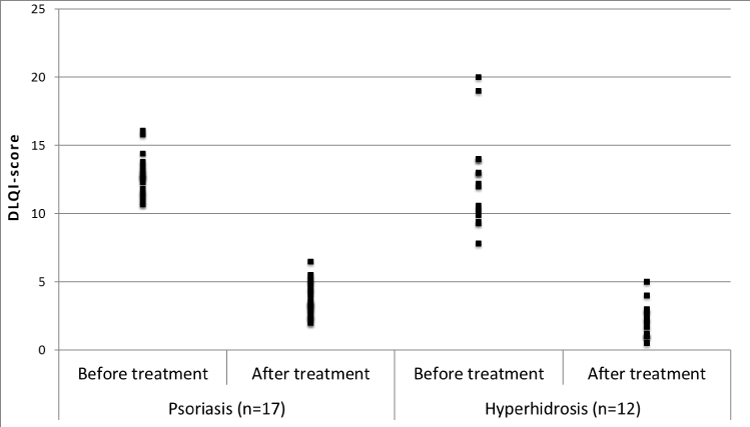
Figure 3: Compilation of the Dermatology Life Quality Index (DLQI) in studies of psoriasis (n = 17) before and after treatment with biological medicines and hyperhidrosis (n = 12) before and after treatment with botulinum toxin. DLQI is a measure of the quality of life where points above 10 are considered to stand for a very large effect on the quality of life8. The compilation includes articles from PubMed, available in full text at Uppsala University Library. Mean values of DLQI have been presented in all of the studies concerning psoriasis, whereas mean values or median values have been presented in the studies concerning hyperhidrosis. The figure is reprinted with permission from Ugeskrift for Læger9.
Patient stories may illustrate the difficulties experienced with hyperhidrosis in different places (Table 2).
| Armpits |
| 22 year-old woman |
| “I cannot have whichever clothes I want. Always have to plan what I am going to wear. It is usually black, loose-fitting jumpers. I take extra jumpers of the same colour with me so I can change at work. Sometimes I insert bandages under my armpits to prevent stains. My movement patterns are affected. Am afraid of lifting my arms. I didn’t put up my hand at school!” |
| Hands |
| 12 year-old boy |
| “When was the first time you noticed that your hands were sweating?” “It was at nursery and we were to walking in pairs. My friend didn’t want to hold my hand.” “What troubles you the most now?” “Everything I touch becomes wet or the fact that my pen slips in my hand.” “Is there anything you avoid doing because your hands sweat?” “If there’s a dance or something on at school I don’t go.” “Have you or your parents spoken to the school about your problems?” “No, I don’t want to.” |
| Feet |
| 34 year-old man |
| “My shoes are destroyed in a month and then have to be thrown away because they smell bad. I don’t like going to people’s homes on the spur of the moment without having extra socks. It’s embarrassing making marks on the parquet flooring and I don’t want to take off my shoes at work due to a bad smell, but at the same time I sweat more when my feet are enclosed. It’s a catch 22. Once my feet were close to freezing because I continued to sweat although it was cold outside.” |
| Groins/Glutes |
| 22 year-old woman |
| “I wear dark jeans because I’m afraid of wetting my underclothes and trousers through. I refuse to sit on plastic chairs because there is a great risk of stains being left. I don’t want to meet a man because I feel that having sex would be inconceivable.” |
| Torso |
| 60 year-old man |
| “I sweat all over my body but the absolute worst is the sweat from my back and chest. Constantly being wet and sticky is unkempt. In the summer I can wet a jacket through if I don’t plan carefully. In the morning I have as cold a shower as possible to avoid sweating afterwards. I then go to work by car and am always out in good time so I can sweat afterwards and change my jumper. The problem is when there are spontaneous meetings or you have to hurry away. That’s when I can no longer plan.” |
| Face/Head |
| 75 year-old woman |
| “I was 51 when I started the menopause but my difficulties started 4-5 years ago. It literally ran from my forehead down onto my face with the minimum of exertion. And my hair looked as though I’d just come out of the shower. I went to my gynaecologist and was given hormones which I’ve been taking up until now, although they don’t actually help. Can’t go shopping, vacuum or go for a walk without it running. In the summer all I have to do is sit still for it to start. My life feels hopeless.” |
| 45 year-old man |
| “People think I’m uncertain and nervous when my face sweats but I don’t feel uncertain - quite the opposite. My mother and grandmother had the same difficulties with sweating from their head. I’ve been treated with various psychotropics and have been for therapy but it hasn’t worked. I’ve now been promoted to head of department but have declined because I’d be much more at the centre of attention. It quite simply feels bloody awful!” |
Table 2: Typical patient accounts from those visiting the Hidrosis Clinic, Stockholm. With validated instruments such as DLQI to measure quality of life, the patients’ suffering can be objectivised, treatments can be assessed and results can be related to other diseases (see Figure 3). The table is reprinted with permission from Läkartidningen2.
Secondary Hyperhidrosis
Secondary hyperhidrosis may involve several specialties (Table 3). A small amount of anamnestic data is usually enough to differentiate between primary and secondary hyperhidrosis (Table 1), but sometimes anamnesis and status in diagnostics are insufficient and that is when it becomes relevant to take samples and do further examinations.
| Dermatology | ||
| Eccrine nevus | ||
| Idiopathic unilateral focal hyperhidrosis | ||
| Vascular deformities | ||
| Pretibial myxoedema | ||
| Gynecology | ||
| Postmenopausal hyperhidrosis | ||
| Iatrogenic | ||
| Medicines: | ||
| • methadone or other opiates | ||
| • cholinergics such as galantamine | ||
| • SSRIs | ||
| • with several medicines | ||
| Infection | ||
| Brucellosis | ||
| HIV | ||
| Chronic malaria | ||
| TBC | ||
| Endocarditis | ||
| With several infectious diseases | ||
| Surgery | ||
| Compensatory hyperhidrosis after sympathectomy | ||
| Medicine | ||
| Diabetes (hyperhidrosis due to neuropathy or hypoglycemia) | ||
| Endocrine diseases: | ||
| • acromegaly | ||
| • pheochromocytoma | ||
| • hyperthyroidism | ||
| • hypogonadism | ||
| • insulinoma | ||
| Heart failure | ||
| Obesity | ||
| Neurology | ||
| Central or peripheral lesion | ||
| Harlequin syndrome | ||
| Horner’s syndrome | ||
| Compensatory hyperhidrosis | ||
| Ross syndrome | ||
| Parkinson’s | ||
| Polyneuropathies | ||
| Oncology | ||
| Carcinoid | ||
| Lymphoma | ||
| With several malignancies | ||
| Orthopedics | ||
| Hyperhidrosis from amputation stump | ||
| Psychiatry | ||
| Anxiety disorder | ||
| Psychotropic drugs | ||
| Social phobia | ||
| Ear, nose and throat | ||
| Frey’s syndrome | ||
Table 3: Secondary hyperhidrosis involves diagnoses from several specialities. In the table follow examples of causes of secondary hyperhidrosis which are important to know of when assessing patients with hyperhidrosis. The table is reprinted with permission from Läkartidningen2.
Secondary general hyperhidrosis
With general hyperhidrosis it can be difficult to clarify whether it is primary or secondary. A long anamnesis with no signs of other disease strongly suggests that it is not an endocrine, infectious or malignant disease.
If there is a short anamnesis you ask whether there are B symptoms, medication has recently been started, there are signs of endocrine disease or the menopause has started. If there are no signs of disease but there is a short anamnesis, a smaller screening is recommended with SR, CRP, blood status, liver and thyroid tests, IGF 1 (acromegaly), metanephrines in plasma (pheochromocytoma) and a lung x-ray. If there are B symptoms other than sweating in the anamnesis, additional and more targeted examinations take place.
In men, low levels of testosterone can lead to general hyperhidrosis but other symptoms such as loss of libido or erection problems can provide diagnostic guidance10.
Obesity is rarely the reason for general hyperhidrosis but can be a deteriorative factor.
Several medicines can cause increased sweating as a side effect and SSRIs and opioids are the most commonly reported. Of the opioids, methadone is particularly problematic.
Polyneuropathies where the sudomotor nerves are damaged cause less sweating from the extremities and compensatory sweating from the head and torso. This compensatory sweating can be misinterpreted as primary general hyperhidrosis. The polyneuropathy can be known as it is in a diabetic but can also be discovered during an examination and must then be investigated and treated if possible.
Sweating during the day and at night is a sub-symptom of the menopause in many women. While other climacteric symptoms disappear, the sweating continues in a significant share of women. As many as 10 per cent of all women suffer from postmenopausal hyperhidrosis 10 years after the menopause11.
Sweating with anxiety disorder or social phobia can be explained by sudden, powerful activation of the sympathetic nerves. Twenty years of experience of hyperhidrosis patients have taught us that hyperhidrosis is rarely primarily due to anxiety disorder; on the other hand, hyperhidrosis can lead to anxiety, palpitations, and escape behaviour. The psychiatric disorder for patients with DSM IV-diagnosed generalised anxiety (SAD) and axillary hyperhidrosis improved following the elimination of the sweat from their armpits with botulinum toxin12. It means that patients with anxiety symptoms and hyperhidrosis should of course receive early help with their somatic disorder.
Secondary Regional/asymmetrical Hyperhidrosis
Regional or asymmetrical sweating is a strong indication of secondary hyperhidrosis and an underlying diagnosis should be sought. Loss of sweating from one area of the body can cause increased sweating from another. This “compensatory” hyperhidrosis was highlighted in the Swedish mass media in the 1990s when the more common sympathectomise for hyperhidrosis were debated. The hands of the patients who underwent the operation certainly became dry but the majority instead sweated below the nipples. Many of these iatrogenically-damaged patients can now receive help with botulinum toxin, sometimes combined with anticholinergics (see case description).
Horner's syndrome with loss of sweating from one side of the face can cause compensatory hyperhidrosis from the contralateral side. With regional hyperhidrosis, it is important to examine the contralateral side with regard to deficit symptoms!
Unusual asymmetrical sweating is seen with idiopathic unilateral focal hyperhidrosis. Attacks of profuse sweating occur from a delimited area, usually on the forehead or the upper side of one wrist. The patient perceives this as very troublesome. There is nothing to note from the outcome of a neurological examination but PAD from a skin biopsy can show a picture that is compatible with eccrine nevus13.
Case Description. Post-Sympathectomy Compensatory Hyperhidrosis
“66 year-old man who has had compensatory sweating for many years following a sympathectomy in the 1970s. Following minor exertion such as walking to catch the bus or vacuuming, the sweat runs from his torso below his nipples. Sometimes the sweat comes while he is sitting still for no actual reason. Has never had a really dry torso or armpits since the operation in the 1970s. Went to the Hidrosis Clinic, Stockholm”.
Was given botulinum toxin injections in the armpits and torso. At the first return appointment 3 weeks after the injections, the patient showed a clear improvement. DLQI fell from 23 to 11. Was started on anticholinergics and, at a return appointment one week later, the patient was completely dry! DLQI had fallen further to 1. The patient was now able to exert himself without his torso sweating. The case description is reprinted with permission from Läkartidningen2.

Image 1: At the first appointment at the Hidrosis Clinic, Stockholm, the patient’s jumper was wet below the denervated area. Normally, the thoracic ganglia 2/3 (and 4) were sympathectomied. This means dry above and wet below the nipples.

Image 2: Following treatment with botulinum toxin and anticholinergics, the patient’s torso was completely dry, even after exertion.
Night Sweats
Many patients describe night sweats as pretty distressing. Wet bedclothes disturb the night’s sleep. Night sweats can be associated with the menopause, infection, malignancy or endocrine disease. At the same time, it is not uncommon for night sweats to occur without a serious underlying cause14. Fluctuating sleep depth due to sleep apnoea, pains, restless legs, etc., seem to affect thermoregulation. The cause of sleep apnoea with night sweats should be investigated15. In our experience, some patients who have night sweats alongside pains or nightmares with superficial, fragmented sleep as a consequence often have a good effect from 10-30 mg amitriptyline at night. Anticholinergics with a long half-life, such as 2 mg tolterodine at night, may also be of value.
Food-triggered (Gustatory) Sweating
Around the mouth and nose there is an evolutionary residue of parasympathetic fibres which innervate sweat glands. The physiological gustatoric reflex involves sensory fibres from the tongue (trigeminus) which are connected to the pterygopalatine ganglion and parasympathetic fibres which innervate oronasal sweat glands. Everyone knows that your face sweats when you consume highly spiced food.
Pathological gustatory hyperhidrosis is due to sympathetic denervation of the sweat glands in the cheek with reinnervation from the parasympathetic fibres which normally innervate oronasal salivary glands and/or sweat glands. This gustatory hyperhidrosis is seen following salivary gland surgery and is then called Frey’s syndrome or following sympathectomies. Damage to sympathetic sudomotor nerves may also be due to diseases like diabetic neuropathy or intrathoracic expansive processes with gustatory hyperhidrosis as a consequence.
Assessment of the Patient
Diagnose conditions underlying secondary hyperhidrosis. If the anamnesis indicates primary hyperhidrosis, no samples need to be taken (table 1).
Is there a connection with starting a medicine? Is there a connection with an operation? Sympathectomy with compensatory hyperhidrosis or salivary gland surgery with subsequent food-triggered (gustatory) sweating of the face (Frey’s syndrome)?
If there is a B symptom, do a targeted investigation specifically for malignancy and infection. If there is denervation, compensatory sweating should be suspected. If there is asymmetrical sweating, a neurological cause should always be suspected. Examine the hyperhidrotic side and the contralateral where it is not rare for it to be anhidrotic (e.g. with Horner’s syndrome).
Understand the patient’s needs. Look at all locations by which the patient is troubled and then prioritise the locations that disrupt the patient the most. Night sweats may be a big problem for some and must also be asked about. When the patient’s prioritations have been highlighted, set up a treatment plan; which areas can be treated with botulinum toxin? If there are remaining areas which cannot be treated with botulinum toxin, anticholinergics can be started. If there are night sweats, these should also be treated if possible.
The benefits and drawbacks of various therapies should be stated before treatment.
Treatment Overview
Depending on the localization of hyperhidrosis, there are varying treatment options. The treatment should be individually adapted with respect to contraindications and therapy failure etc. The availability of drugs can differ between countries due to lack of approval from the authorities. Various treatment options are described below and Table 4 displays recommendations based on the clinical guidelines from International Hyperhidrosis Society16, recommendations from the Multi-Specialty Working Group of the Recognition, Diagnosis and Treatment of Primary Focal Hyperhidrosis17 and Botulinum Toxin Treatment of Autonomic Disorders: Focal Hyperhidrosis and Sialorrhea18.
Topical Treatment
Topical treatment, e.g. aluminum chloride (AlCl), should be tried on all localised hyperhidrosis16,17. Application may however be difficult on the scalp because of hairy skin. AlCl reacts with proteins in the sweat duct and forms a mechanical obstacle which prevents sweating. Solution is applied on completely dry skin once a week or more, preferably when going to bed and leave to work overnight. Some patients have problems using the solution due to skin irritation.
Botulinum toxin type A (BTX A)
Intradermal, local injections with BTX A constitutes a very effective treatment. Major, randomised multicentre studies have taken place concerning the indication of axillary hyperhidrosis19,20 and several studies have also shown a good effect for other locations21-29. BTX A causes local chemical denervation by preventing the release of acetylcholine. The duration of the effect differs between individuals and the treatment usually needs to be repeated 1-4 times a year. Most commonly reported side effects are dryness of the skin and slight muscular weakness. Any side effects are usually local, transient and mild.
Botulinum Toxin type B (BTX B)
The mechanism of action for BTX B is similar to that for BTX A but the effect on α-motor neurons to muscles seems to be much less because 50-100 times higher doses are needed to treat cervical dystonia. However, new research shows that BTX B can be diluted to a low concentration to a greater extent and that the sweat-inhibiting effect of 1 U BTX A corresponds to 1-2 U BTX B30,31.
These research results have generated possibilities of administering treatment to large areas or areas where there is a great risk of local muscular side effects, as in the palms or the face32,33. Patients with craniofacial hyperhidrosis33, general hyperhidrosis or postmenopausal hyperhidrosis where oestrogen by mouth is unsuitable or ineffectual can receive treatment with relatively small doses of BTX B. The hands of children, musicians or others where local muscular weakness is not acceptable can also be treated with BTX B with a small risk of side effects34. On group basis the duration of effect might be a bit shorter after treatment with BTX B compared with BTX A, but not for all individuals.
Iontophoresis
Iontophoresis may be an alternative in the treatment of palmar/plantar hyperhidrosis. Ions are conveyed to the sweat ducts using a weak current and cause an obstacle in the outermost section of the sweat duct. Initially, the treatment is administered 3-4 times per week for 20-30 minutes on each occasion. The interval is then lengthened and individually adapted with 1-4 treatments usually required per month. The treatment is time-consuming for the patient.
Microwave thermolysis
In recent years, a non-invasive method which causes local destruction of sweat glands through microwaves (miraDry®) has been developed with satisfactory and permanent results35. Other invasive or minimally-invasive methods are available for isolated axillary hyperhidrosis if miraDry® is contraindicated.
Systemic medications
Different types of oral anticholinergic medications can work well for multifocal hyperhidrosis and alongside BTX, lead to additive effects. Monotherapy generally has an inadequate effect. Due to the systemic impact, the risk of side effects is greater compared with local treatment. Dry mouth is very commonly reported, furthermore, urinary retention, dry eyes and accommodation disturbance occur. Indications of an increased risk of dementia where there is higher cumulative use of anticholinergic medications has led to a demand for greater awareness of the potential risks of the use of anticholinergics and to reduce their use over time (36). In small studies, peroral preparations such as calcium channel blockers or carbonic anhydrase inhibitors (direct effect on the sweat glands calcium channels or carbonic anhydrase) and clonidine have proven to have an effect and can be tried.
Endoscopic Thoracic Sympathectomy (ETS)
Owing to the frequency of severe and irreversible side effects, ETS is no longer performed in Sweden. The side effects profile is unfavourable with acute and chronic side effects. The most common of reported side effects is compensatory sweating (incidence of 80-95 % has been reported in several studies) and may constitute a lifelong disability, which for many is a significantly greater disorder than that which justified the operation (see case description)37,38.
In countries where ETS still is performed, patients must be carefully selected and educated to fully understand the possibility of limited efficacy and the risks of complications including, but not limited to, compensatory sweating16. This treatment should be the last option16,17.
| Option | Axillary hyperhidrosis | Palmar hyperhidrosis | Plantar hyperhidrosis | Craniofacial hyperhidrosis | Gustatory hyperhidrosis | Torso (compensatory hyperhidrosis) |
|---|---|---|---|---|---|---|
| 1st | Topical treatment (e.g. AlCl) |
Topical treatment (e.g. AlCl) / Iontophoresis |
Topical treatment (e.g. AlCl) / Iontophoresis |
Topical treatment (e.g. AlCl)† |
Topical treatment (e.g. AlCl) / Botulinum toxin |
Botulinum toxin |
| 2nd | Botulinum toxin / microwave thermolysis | Botulinum toxin | Botulinum toxin | Botulinum toxin | Botulinum toxin / Topical treatment (e.g. AlCl) |
Systemic medications (e.g. anticholinergics) |
| 3rd | Local sweat gland ablation | Systemic medications (e.g. anticholinergics) | Systemic medications (e.g. anticholinergics) | Systemic medications (e.g. anticholinergics) | Systemic medications (e.g. anticholinergics) | |
| 4th | Systemic medications (e.g. anticholinergics) | (ETS)* | (ETS)* | |||
| 5th | (ETS)* |
Table 4: Treatment recommendations based on the clinical guidelines from International Hyperhidrosis society16, recommendations from the Multi-Specialty Working Group of the Recognition, Diagnosis and Treatment of Primary Focal Hyperhidrosis17 and Botulinum Toxin Treatment of Autonomic Disorders: Focal Hyperhidrosis and Sialorrhea.18.
† Application on the scalp may be difficult because of hairy skin.
*In countries where Endoscopic Thoracic Sympathectomy (ETS) still is performed, patients must be carefully selected and educated to fully understand the possibility of limited efficacy and the risks of complications including, but not limited to, compensatory sweating16.
Conclusion
Hyperhidrosis is a widespread disorder which is usually idiopathic but can be secondary to diseases involving several specialities. Examinations show that there is a strong negative impact on the quality of life of patients with hyperhidrosis. With today’s treatment methods, individually-adapted therapy can lead to very good results.
References
- Schlereth T, Dieterich M, Birklein F. Hyperhidrosis--causes and treatment of enhanced sweating. Deutsches Arzteblatt international. 2009 Jan;106(3):32-7.
- Swartling C, Brismar K, Aquilonius SM, Naver H, Rystedt A, Rosell K. [Hyperhidrosis--the "silent" handicap]. Lakartidningen. 2011 Nov 23-29;108(47):2428-32.
- Grammer K, Fink B, Neave N. Human pheromones and sexual attraction. European journal of obstetrics, gynecology, and reproductive biology. 2005 Feb 1;118(2):135-42.
- Iwase S, Ikeda T, Kitazawa H, Hakusui S, Sugenoya J, Mano T. Altered response in cutaneous sympathetic outflow to mental and thermal stimuli in primary palmoplantar hyperhidrosis. Journal of the autonomic nervous system. 1997 Jun 6;64(2-3):65-73.
- Strutton DR, Kowalski JW, Glaser DA, Stang PE. US prevalence of hyperhidrosis and impact on individuals with axillary hyperhidrosis: results from a national survey. Journal of the American Academy of Dermatology. 2004 Aug;51(2):241-8.
- Kaufmann H, Saadia D, Polin C, Hague S, Singleton A, Singleton A. Primary hyperhidrosis--evidence for autosomal dominant inheritance. Clin Auton Res. 2003 Apr;13(2):96-8.
- Kim WO, Kil HK, Yoon KB, Yoo JH. Treatment of generalized hyperhidrosis with oxybutynin in post-menopausal patients. Acta dermato-venereologica. 2010 May;90(3):291-3.
- Hongbo Y, Thomas CL, Harrison MA, Salek MS, Finlay AY. Translating the science of quality of life into practice: What do dermatology life quality index scores mean? The Journal of investigative dermatology. 2005 Oct;125(4):659-64.
- Swartling C, Rystedt A, Brismar K, Naver H, Norman P, Dizdarevic A. [Hyperhidrosis is a silent handicap]. Ugeskrift for laeger. 2016 Feb 8;178(6):V04150291.
- Vermeulen A. Diagnosis of partial androgen deficiency in the aging male. Annales d'endocrinologie. 2003 Apr;64(2):109-14.
- Berg G, Gottwall T, Hammar M, Lindgren R. Climacteric symptoms among women aged 60-62 in Linkoping, Sweden, in 1986. Maturitas. 1988 Oct;10(3):193-9.
- Connor KM, Cook JL, Davidson JR. Botulinum toxin treatment of social anxiety disorder with hyperhidrosis: a placebo-controlled double-blind trial. The Journal of clinical psychiatry. 2006 Jan;67(1):30-6.
- Kreyden OP, Schmid-Grendelmeier P, Burg G. Idiopathic localized unilateral hyperhidrosis: case report of successful treatment with botulinum toxin type A and review of the literature. Archives of dermatology. 2001 Dec;137(12):1622-5.
- Mold JW, Roberts M, Aboshady HM. Prevalence and predictors of night sweats, day sweats, and hot flashes in older primary care patients: an OKPRN study. Annals of family medicine. 2004 Sep-Oct;2(5):391-7.
- Kiely JL, Murphy M, McNicholas WT. Subjective efficacy of nasal CPAP therapy in obstructive sleep apnoea syndrome: a prospective controlled study. Eur Respir J. 1999 May;13(5):1086-90.
- International Hyperhidrosis Society. Algorithms hyperhidrosis. http://www.sweathelp.org/treatments-hcp/clinical-guidelines.html [2016-06-21].
- Hornberger J, Grimes K, Naumann M, Glaser DA, Lowe NJ, Naver H, et al. Recognition, diagnosis, and treatment of primary focal hyperhidrosis. Journal of the American Academy of Dermatology. 2004 Aug;51(2):274-86.
- Hosp C, Naumann MK, Hamm H. Botulinum Toxin Treatment of Autonomic Disorders: Focal Hyperhidrosis and Sialorrhea. Seminars in neurology. 2016 Feb;36(1):20-8.
- Heckmann M, Ceballos-Baumann AO, Plewig G. Botulinum toxin A for axillary hyperhidrosis (excessive sweating). The New England journal of medicine. 2001 Feb 15;344(7):488-93.
- Naumann M, Lowe NJ. Botulinum toxin type A in treatment of bilateral primary axillary hyperhidrosis: randomised, parallel group, double blind, placebo controlled trial. BMJ (Clinical research ed. 2001 Sep 15;323(7313):596-9.
- Schnider P, Binder M, Auff E, Kittler H, Berger T, Wolff K. Double-blind trial of botulinum A toxin for the treatment of focal hyperhidrosis of the palms. The British journal of dermatology. 1997 Apr;136(4):548-52.
- Lowe NJ, Yamauchi PS, Lask GP, Patnaik R, Iyer S. Efficacy and safety of botulinum toxin type a in the treatment of palmar hyperhidrosis: a double-blind, randomized, placebo-controlled study. Dermatol Surg. 2002 Sep;28(9):822-7.
- Campanati A, Bernardini ML, Gesuita R, Offidani A. Plantar focal idiopathic hyperhidrosis and botulinum toxin: a pilot study. Eur J Dermatol. 2007 Jan-Feb;17(1):52-4.
- Vadoud-Seyedi J. Treatment of plantar hyperhidrosis with botulinum toxin type A. International journal of dermatology. 2004 Dec;43(12):969-71.
- Hexsel DM, Dal'forno T, Hexsel CL. Inguinal, or Hexsel's hyperhidrosis. Clinics in dermatology. 2004 Jan-Feb;22(1):53-9.
- Kim WO, Kil HK, Yoon KB, Noh KU. Botulinum toxin: a treatment for compensatory hyperhidrosis in the trunk. Dermatol Surg. 2009 May;35(5):833-8; discussion 8.
- Ferraro G, Altieri A, Grella E, D'Andrea F. Botulinum toxin: 28 patients affected by Frey's syndrome treated with intradermal injections. Plastic and reconstructive surgery. 2005 Jan;115(1):344-5
- Kinkelin I, Hund M, Naumann M, Hamm H. Effective treatment of frontal hyperhidrosis with botulinum toxin A. The British journal of dermatology. 2000 Oct;143(4):824-7.
- Naver H, Swartling C, Aquilonius SM. Treatment of focal hyperhidrosis with botulinum toxin type A. Brief overview of methodology and 2 years’ experience. Eur J Neurol. 1999;6 (suppl 4):S117-20.
- Rystedt A, Swartling C, Naver H. Anhidrotic effect of intradermal injections of botulinum toxin: a comparison of different products and concentrations. Acta dermato-venereologica. 2008;88(3):229-33.
- Rystedt A, Karlqvist M, Bertilsson M, Naver H, Swartling C. Effect of botulinum toxin concentration on reduction in sweating: a randomized, double-blind study. Acta dermato-venereologica. 2013 Nov;93(6):674-8.
- Swartling C, Farnstrand C, Abt G, Stalberg E, Naver H. Side-effects of intradermal injections of botulinum A toxin in the treatment of palmar hyperhidrosis: a neurophysiological study. Eur J Neurol. 2001 Sep;8(5):451-6.
- Karlqvist M, Rosell K, Rystedt A, Hymnelius K, Swartling C. Botulinum toxin B in the treatment of craniofacial hyperhidrosis. J Eur Acad Dermatol Venereol. 2013 Oct 3.
- Rosell K, Hymnelius K, Swartling C. Botulinum toxin type a and B improve quality of life in patients with axillary and palmar hyperhidrosis. Acta dermato-venereologica. 2013 May 2;93(3):335-9.
- Hong HC, Lupin M, O'Shaughnessy KF. Clinical evaluation of a microwave device for treating axillary hyperhidrosis. Dermatol Surg. 2012 May;38(5):728-35. PubMed PMID: 22452511.
- Gray SL, Anderson ML, Dublin S, Hanlon JT, Hubbard R, Walker R, et al. Cumulative use of strong anticholinergics and incident dementia: a prospective cohort study. JAMA internal medicine. 2015 Mar;175(3):401-7. PubMed PMID: 25621434.
- Furlan AD, Mailis A, Papagapiou M. Are we paying a high price for surgical sympathectomy? A systematic literature review of late complications. The journal of pain : official journal of the American Pain Society. 2000 Winter;1(4):245-57.
- Walles T, Somuncuoglu G, Steger V, Veit S, Friedel G. Long-term efficiency of endoscopic thoracic sympathicotomy: survey 10 years after surgery. Interactive cardiovascular and thoracic surgery. 2009 Jan;8(1):54-7
