Membrane Remodelling Activity of α-Synuclein
Silvia Campioni1,2, Roland Riek1*
Abstract
Despite extensive research, a detailed description of the physiological function of α-Synuclein (α-Syn), the human neuronal protein involved in the pathogenesis of Parkinson’s Disease, is still lacking, most likely due to its highly dynamic conformation and behaviour. Recently, it has become increasingly evident that the interaction of α-Syn with membranes plays an important role in its function and misfunction. Strikingly, despite not having a membrane scaffolding domain, α-Syn can extensively reshape membrane bilayers. Moreover, stable and soluble nanometer-sized particles, whose morphology is ranging from tubules to discoids, can be obtained in vitro with different protocols and from different lipids. The focus of this review article is on the description of the membrane remodelling activity of α-Syn and on its possible physiological role.
Text
Human α-Syn is a neuronal protein, a member of the synuclein protein family, and the major component of the abnormal proteinaceous inclusions termed Lewy bodies detected in the brain of individuals affected by Parkinson’s Disease (PD), dementia with Lewy Bodies and multiple system atrophy1. In addition to its disease association, considerable research effort is nowadays dedicated to understand the functional role of α-Syn in the brain2,3. α-Syn is highly abundant, estimated to account for 0.5-1% of the total protein content in the cytosol of human brain homogenates4. The synuclein name originates from its localization at the presynaptic terminals and at the nuclear envelope of cholinergic neurons of Torpedo californica5. While the detection of nuclear α-Syn has often failed, its presynaptic localization has been confirmed also in neurons from other regions of the brain and from other organisms, including humans4,6-8. A recent semi-quantitative analysis of the subcellular localization of α-Syn in neurons from different regions of rat brains indicates that it is present in almost all subcellular fractions, but its density is particularly high at presynaptic terminals, at the nuclear envelope, and in mitochondria9. It is further important to mention, that α-Syn is present not only in the central nervous system but also peripherally, including the enteric nervous system.
From a structural point of view, α-Syn is highly plastic and dynamic10. When free in solution or in the intracellular milieu, this protein adopts a largely disordered conformation, which is devoid of stable secondary structure elements, but still more compact than the one expected for a random coil state11-14. Upon prolonged incubation in test tubes (most often with shaking at high concentration and in presence of an air/water interface) α-Syn has been reported to aggregate into fibrils with the morphological, tinctorial and structural properties of amyloids15-19. Finally, when lipid vesicles are added to solutions containing monomeric and disordered α-Syn, a transition to a helix-rich state is observed12,20. The binding of α-Syn to lipids and detergents (particularly anionic ones) is mediated by seven copies of an imperfect 11-amino acid residues-long repeat (XKTKEGVXXXX) that resembles the one seen in the amphipathic helices of apolipoproteins6,21,22. The capability of α-Syn to bind to membranes is supposed to play an important role in both its function and aggregation. At presynaptic terminals, α-Syn is loosely associated to the distal (or reserve) pool of synaptic vesicles, suggesting its involvement in the regulation of synaptic vesicles release and recycling8,9,23. However the physiological function of α-Syn remains elusive24-26. In this article, we will review the membrane remodelling activity of α-Syn, also in light of the recent observation that stable tubular or discoidal nanoparticles consisting of α-Syn and phospholipids can be formed in vitro27-34. We will discuss the features of these species and make hypothesis on the physiological relevance of the membrane remodelling activity exerted by α-Syn.
α-Syn Induces Membrane Tubulation
The flexible and dynamic nature of α-Syn is also reflected by the fact that multiple binding modes to membranes have been revealed20,35,36. The helical conformation adopted by the first 100 amino acid residues of α-Syn upon binding can be extended and uninterrupted37,38, or consist of two helices connected by a short loop in a horseshoe-like fold39,40. Even if both conformers appear to co-exist and undergo interconversion36,41,42, the broken conformation seems to be preferred on highly curved lipid surfaces. The curvature of the membrane also affects the extent of α-Syn binding, as this protein preferably binds to small, hence highly curved, vesicles22,43,44. In addition to the capability to sense membrane curvature, α-Syn can also induce extensive membrane reshaping upon binding, even if its sequence does not contain a membrane scaffolding domain like those of known curvature-inducing proteins involved in membrane remodelling and vesicle trafficking (e.g. endophilin and amphiphysin)45,46. In particular, when monomeric α-Syn is added to turbid suspensions of giant lipid vesicles containing negatively charged phospholipids, the sample becomes rapidly clear27. Depending on the lipid composition and on the protein to lipid ratio, different particles are generated, in particular long tubules with varying diameter and circular structures resembling vesicles with the α-Syn protein laying at their surface27. Similar structures are also observed in vitro when amphiphysin is added to the same type of vesicles; moreover, apolipoprotein A-I is also capable to induce the tubulation of giant vesicles, although it prefers less negatively charged ones27. Lipid tubules form rapidly even upon addition of monomeric α-Syn to supported lipid bilayers containing anionic phopsholipids28. At fixed lipid composition, the measured diameter of the tubules generated by α-Syn varies with the protein-to-lipid ratio, with larger tubules being more abundant at lower ratios29. Changes in the length and bulkiness of the lipid acyl chain determine the nature of the tubules: with short chain phospholipids only cylindrical micelles observed; with phospholipids having longer and bulkier acyl chains bilayer tubes become apparent and in some cases even more abundant than the cylindrical micelles29.
The binding of α-Syn to giant lipid vesicles causes membrane expansion33, an effect which has also been observed by addition of α-Syn to supported lipid bilayers47. A possible mechanism of membrane remodelling, based on atomic force microscopy images, is that α-Syn binding induces lipid interdigitation and their lateral expansion out of the plane of the membrane, leading to bilayer thinning47. Determining at high-resolution the structure of membrane bound α-Syn and its interplay with lipids appears challenging. In a recent solid-state solution state nuclear magnetic resonance (NMR) study of α-Syn on small vesicles mimicking synaptic ones, only the segment residues 6-25 could be assigned48, while with solution-state NMR multiple lipid binding modes were elucidated with the 40 C-terminal residues being dynamically disordered20,48. On cylindrical micelles, electron paramagnetic resonance spectroscopy (EPR)29 data indicate that α-Syn adopts an ordered structure (but devoid of quaternary contacts), having the first 100 amino acid residues in contact with the membrane, while the rest of the sequence remains disordered. The measured EPR spectra are similar to those of vesicle-bound α-Syn, but completely different from those of fibrillar α-Syn indicating that lipid tubulation (which is anyway occurring on a much faster time scale than amyloid formation) is not caused by the fibrillar state of α-Syn, but rather by monomeric or small oligomeric species in a helical conformation. Distance measurements between different pairs of labelled residues in the membrane-bound region suggest that the helical state adopted at the level of the tubules is extended29.
α-Syn forms Stable Lipoprotein Nanoparticles
At high protein to lipid ratios (at least 1:10), negatively charged giant lipid vesicles are remodelled into discoidal or oval nanoparticles having a diameter of 6.5-10 nm29,30. These nanoparticles are stable and they can be isolated from tubular structures by centrifugation and subsequent size-exclusion chromatography30. Their size and morphology is similar to that of lipoprotein particles formed by other proteins containing highly conserved 11-mer amino acid sequence repeats that form amphipathic helices (e.g. apolipoproteins and perilipins). Incubation of α-Syn with small unilamellar vesicles (diameter of ca. 25 nm) also results in the formation of nanoparticles undistinguishable from those obtained from the giant vesicles30. Notably, membrane scaffolding proteins (MSPs), whose sequence is based on that of apoliproproteins, are routinely used in vitro to prepare soluble nanoscale phospholipid bilayers, termed nanodiscs, that serve to investigate the structure and function of membrane proteins49,50. When a protocol analogous to that used to prepare nanodiscs, but with α-Syn replacing MSPs, is adopted, homogeneous and stable nanoparticles having a diameter of 19-28 nm32 were obtained. With this method, a low protein-to-lipid radio (1:40) is already sufficient to get full incorporation of monomeric α-Syn into nanoparticles composed of negatively charged phospholipids. In addition, albeit at lower yield, it is possible to obtain lipoparticles of similar size also with zwitterionic sphingomyelin (a lipid component of synaptic vesicles), while no nanoparticles could be generated by addition of monomeric α-Syn to giant vesicles made of zwitterionic phosphatidylcholine30,32. Albeit bigger, the nanoparticles obtained with the nanodisc-like protocol appear to have features similar to those obtained in vesicle remodelling experiments: in both cases, α-Syn adopts a helical conformation involving the first 100 amino acid residues of its sequence and cryo-electron microscopy densities indicate that it wraps around the nanoparticles in a ring-like manner, an architecture similar to the one reported for the early discoidal high density lipoprotein particles formed by apolipoprotein A-151. Moreover, α-Syn oligomers are present in both 6.5-10 nm (dimers to tetramers) and 19-28 nm (octamers to decamers) particles, their size consistently varying with that of the particle30,32. The protein to lipid ratio in the nanoparticles has been estimated to be ca. 1:20-25 for the 6.5-10 nm particles and 1:8-10 for the 19-28 nm one, respectively. In contrast with the results obtained from tubules, EPR data show that in the 6.5-10 nm nanoparticles α-Syn adopts a broken helical state, with the second helix still partially disordered30. This difference might be due to the fact that these nanoparticles are too small to accommodate the extended helix.
Physiological Relevance of the Membrane Remodelling Activity of α-Syn
With the present knowledge on the membrane remodelling activity of α-Syn, it becomes intriguing to make a hypothesis on its physiological significance. Recently, it has been shown that all three human synucleins, α, β and γ 52, induce the tubulation of vesicles composed of brain polar lipids, at concentrations estimated to be physiologically relevant31. In vitro, the remodelling activity is most efficient when pure negatively charged membranes are involved27,30,32,53. The fact that α-Syn shares common features with known curvature-inducing protein and with apolipoproteins suggests shared functionalities and a direct involvement of α-Syn in vesicle trafficking and lipid transport/storage. It is also worth noticing that in triple knockout mice, lacking all three synucleins, altered expression levels of four proteins involved in curvature sensing and generation have been measured, suggesting that their up-regulation is needed to compensate the loss of synucleins31. As mentioned in the Introduction, α-Syn is highly abundant in neurons and its similarity to the afore-mentioned proteins suggests a structural role for this protein. In addition, a role of α-Syn in brain lipid metabolism has been proposed54. A sound hypothesis is therefore that α-Syn may serve to temporarily store negatively charged lipids in soluble lipoprotein particles as a source of negatively charged lipids in the remodelling events that take place at membranes (Figure 1). The schematic drawing of Figure 1 shows that the negatively charged lipids are surrounded by α-Syn forming a lipid container with architecture reminiscent of high density lipoprotein (HDL) particles. Furthermore, the envisioned lipid release from the lipid container to the synaptic membrane is illustrated.
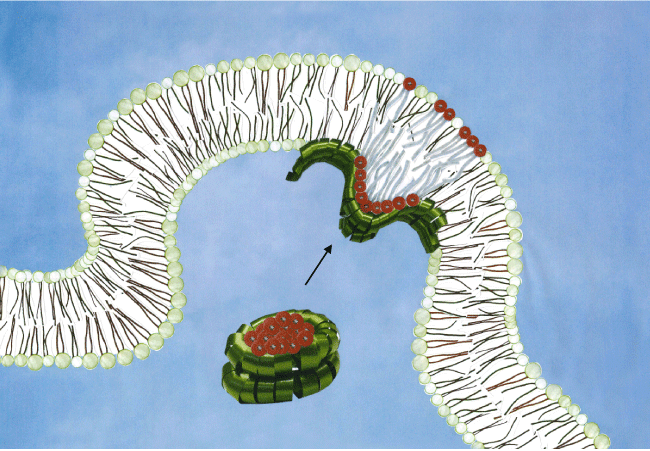
Figure 1:Schematic of the hypothesized role that α-Syn is a storage entity of negatively charged lipids. A α-Syn lipoprotein particle composed of green-colored α-Syn (i.e. decorative ribbon) surrounding negatively charged lipids with red-colored head groups is shown at a synapse. The lipid bilayer is represented by lipids having a non-charged spherical head group. negatively charged lipids. It is envisioned, that this process is initiated upon synapse activation or/and remodelling in concert with other proteins.
In addition, at synapses the membrane remodelling activity of α-Syn may help stabilizing synaptic vesicles and mediate their exo-/endocytosis. Support of this hypothesis is that the expression levels of synucleins and endophilin A1 (a protein involved in endocytosis) appear to be reciprocally regulated and that α-Syn maintains neurotransmitter homeostasis by regulating fusion, clustering, and trafficking of synaptic vesicles55.
Finally, the membrane remodelling activity of α-Syn may also play an important role in the pathogenesis of PD. Lipids are a significant component of the Lewy Bodies inclusions and they have been proposed to derive from degraded membrane organelles. High α-Syn levels cause fragmentation and fusion of mitochondrial membranes, and α-Syn can also remodel giant vesicles having a mitochondrial membrane-like lipid composition30, and mitochondria impairment is an important feature of PD56. Thus, altered intracellular levels of α-Syn may lead to an unbalanced membrane remodelling activity for this protein that might impair cell viability independent of the formation of α-Syn amyloid fibrils.
Conclusion
We have described, to the best of our knowledge, the present information on the membrane remodelling activity of α-Syn. On the basis on this discussed pluripotent feature it is highly likely that it is key for both α-Syn function as well as mis-function in vivo – two big unknowns to be elucidated.
Acknowledgment
We would like to thank Kristina Comiotto for the art work in Figure 1 and the SNF for the financial support through a Synergia grant and a Marie Heim- Voegtlin grant (Number 164425).
References
- Goedert M, Spillantini MG, Del tredici K, Braak H. 100 years of Lewy pathology. Nat Rev Neurol. 2013;9(1):13-24.
- Savica R, Grossardt BR, Bower JH, Ahlskog JE, Rocca WA. Incidence and pathology of synucleinopathies and tauopathies related to parkinsonism. JAMA Neurol. 2013;70(7):859-66.
- Burré J, Sharma M, Tsetsenis T, Buchman V, Etherton MR, Südhof TC. Alpha-synuclein promotes SNARE-complex assembly in vivo and in vitro. Science. 2010;329(5999):1663-7.
- Iwai A, Masliah E, Yoshimoto M, et al. The precursor protein of non-A beta component of Alzheimer's disease amyloid is a presynaptic protein of the central nervous system. Neuron. 1995;14(2):467-75.
- Maroteaux L, Campanelli JT, Scheller RH. Synuclein: a neuron-specific protein localized to the nucleus and presynaptic nerve terminal. J Neurosci. 1988;8(8):2804-15.
- George JM, Jin H, Woods WS, Clayton DF. Characterization of a novel protein regulated during the critical period for song learning in the zebra finch. Neuron. 1995;15(2):361-72.
- Jakes R, Spillantini MG, Goedert M. Identification of two distinct synucleins from human brain. FEBS Lett. 1994;345(1):27-32.
- Kahle PJ, Neumann M, Ozmen L, et al. Subcellular localization of wild-type and Parkinson's disease-associated mutant alpha -synuclein in human and transgenic mouse brain. J Neurosci. 2000;20(17):6365-73.
- Zhang L, Zhang C, Zhu Y, et al. Semi-quantitative analysis of alpha-synuclein in subcellular pools of rat brain neurons: an immunogold electron microscopic study using a C-terminal specific monoclonal antibody. Brain Res. 2008;1244:40-52.
- Uversky VN. A protein-chameleon: conformational plasticity of alpha-synuclein, a disordered protein involved in neurodegenerative disorders. J Biomol Struct Dyn. 2003;21(2):211-34.
- Weinreb PH, Zhen W, Poon AW, Conway KA, Lansbury PT. NACP, a protein implicated in Alzheimer's disease and learning, is natively unfolded. Biochemistry. 1996;35(43):13709-15.
- Eliezer D, Kutluay E, Bussell R, Browne G. Conformational properties of alpha-synuclein in its free and lipid-associated states. J Mol Biol. 2001;307(4):1061-73.
- Fauvet B, Mbefo MK, Fares MB, et al. α-Synuclein in central nervous system and from erythrocytes, mammalian cells, and Escherichia coli exists predominantly as disordered monomer. J Biol Chem. 2012;287(19):15345-64.
- Theillet FX, Binolfi A, Bekei B, et al. Structural disorder of monomeric α-synuclein persists in mammalian cells. Nature. 2016;530(7588):45-50.
- Giehm L, Lorenzen N, Otzen DE. Assays for α-synuclein aggregation. Methods. 2011; 53 (3): 295.
- Campioni S, Carret G, Jordens S, Nicoud L, Mezzenga R, Riek R. The presence of an air-water interface affects formation and elongation of α-Synuclein fibrils. J Am Chem Soc. 2014;136(7):2866-75.
- Conway KA, Harper JD, Lansbury PT. Fibrils formed in vitro from alpha-synuclein and two mutant forms linked to Parkinson's disease are typical amyloid. Biochemistry. 2000;39(10):2552-63.
- Heise H, Hoyer W, Becker S, Andronesi OC, Riedel D, Baldus M. Molecular-level secondary structure, polymorphism, and dynamics of full-length alpha-synuclein fibrils studied by solid-state NMR. Proc Natl Acad Sci USA. 2005;102(44):15871-6.
- Tuttle MD, Comellas G, Nieuwkoop AJ, et al. Solid-state NMR structure of a pathogenic fibril of full-length human α-synuclein. Nat Struct Mol Biol. 2016;23(5):409-15.
- Bodner CR, Dobson CM, Bax A. Multiple tight phospholipid-binding modes of alpha-synuclein revealed by solution NMR spectroscopy. J Mol Biol. 2009;390(4):775-90.
- Segrest JP, Jones MK, De loof H, Brouillette CG, Venkatachalapathi YV, Anantharamaiah GM. The amphipathic helix in the exchangeable apolipoproteins: a review of secondary structure and function. J Lipid Res. 1992;33(2):141-66.
- Davidson WS, Jonas A, Clayton DF, George JM. Stabilization of alpha-synuclein secondary structure upon binding to synthetic membranes. J Biol Chem. 1998;273(16):9443-9.
- Lee SJ, Jeon H, Kandror KV. Alpha-synuclein is localized in a subpopulation of rat brain synaptic vesicles. Acta Neurobiol Exp (Wars). 2008;68(4):509-15.
- Snead D, Eliezer D. Alpha-synuclein function and dysfunction on cellular membranes. Exp Neurobiol. 2014;23:292-313.
- Bendor JT, Logan TP, Edwards RH. The function of α-synuclein. Neuron. 2013; 79 (6): 1044-66.
- Lashuel HA, Overk CR, Oueslati A, Masliah E. The many faces of α-synuclein: from structure and toxicity to therapeutic target. Nat Rev Neurosci. 2013;14(1):38-48.
- Varkey J, Isas JM, Mizuno N, et al. Membrane curvature induction and tubulation are common features of synucleins and apolipoproteins. J Biol Chem. 2010;285(42):32486-93.
- Pandey AP, Haque F, Rochet JC, Hovis JS. α-Synuclein-induced tubule formation in lipid bilayers. J Phys Chem B. 2011;115(19):5886-93.
- Mizuno N, Varkey J, Kegulian NC, et al. Remodeling of lipid vesicles into cylindrical micelles by α-synuclein in an extended α-helical conformation. J Biol Chem. 2012;287(35):29301-11.
- Varkey J, Mizuno N, Hegde BG, Cheng N, Steven AC, Langen R. α-Synuclein oligomers with broken helical conformation form lipoprotein nanoparticles. J Biol Chem. 2013;288(24):17620-30.
- Westphal CH, Chandra SS. Monomeric synucleins generate membrane curvature. J Biol Chem. 2013;288(3):1829-40.
- Eichmann C, Campioni S, Kowal J, et al. Preparation and Characterization of Stable α-Synuclein Lipoprotein Particles. J Biol Chem. 2016;291(16):8516-27.
- Shi Z, Sachs JN, Rhoades E, Baumgart T. Biophysics of α-synuclein induced membrane remodelling. Phys Chem Chem Phys. 2015;17(24):15561-8.
- Taneva SG, Lee JM, Cornell RB. The amphipathic helix of an enzyme that regulates phosphatidylcholine synthesis remodels membranes into highly curved nanotubules. Biochim Biophys Acta. 2012;1818(5):1173-86.
- Vermaas JV, Tajkhorshid E. Conformational heterogeneity of α-synuclein in membrane. Biochim Biophys Acta. 2014;1838(12):3107-17.
- Lokappa SB, Ulmer TS. Alpha-synuclein populates both elongated and broken helix states on small unilamellar vesicles. J Biol Chem. 2011;286(24):21450-7.
- Jao CC, Hegde BG, Chen J, Haworth IS, Langen R. Structure of membrane-bound alpha-synuclein from site-directed spin labeling and computational refinement. Proc Natl Acad Sci USA. 2008;105(50):19666-71.
- Jao CC, Der-sarkissian A, Chen J, Langen R. Structure of membrane-bound alpha-synuclein studied by site-directed spin labeling. Proc Natl Acad Sci USA. 2004;101(22):8331-6.
- Chandra S, Chen X, Rizo J, Jahn R, Südhof TC. A broken alpha -helix in folded alpha -Synuclein. J Biol Chem. 2003;278(17):15313-8.
- Ulmer TS, Bax A. Comparison of structure and dynamics of micelle-bound human alpha-synuclein and Parkinson disease variants. J Biol Chem. 2005;280(52):43179-87.
- Robotta M, Braun P, Van rooijen B, Subramaniam V, Huber M, Drescher M. Direct evidence of coexisting horseshoe and extended helix conformations of membrane-bound alpha-synuclein. Chemphyschem. 2011;12(2):267-9.
- Georgieva ER, Ramlall TF, Borbat PP, Freed JH, Eliezer D. The lipid-binding domain of wild type and mutant alpha-synuclein: compactness and interconversion between the broken and extended helix forms. J Biol Chem. 2010;285(36):28261-74.
- Middleton ER, Rhoades E. Effects of curvature and composition on α-synuclein binding to lipid vesicles. Biophys J. 2010;99(7):2279-88.
- Pranke IM, Morello V, Bigay J, et al. α-Synuclein and ALPS motifs are membrane curvature sensors whose contrasting chemistry mediates selective vesicle binding. J Cell Biol. 2011;194(1):89-103.
- Frost A, Unger VM, De camilli P. The BAR domain superfamily: membrane-molding macromolecules. Cell. 2009;137(2):191-6.
- Mcmahon HT, Gallop JL. Membrane curvature and mechanisms of dynamic cell membrane remodelling. Nature. 2005;438(7068):590-6.
- Ouberai MM, Wang J, Swann MJ, Galvagnion C, Guilliams T, Dobson CM, et al. α-Synuclein senses lipid packing defects and induces lateral expansion of lipids leading to membrane remodeling. J Biol Chem. 2013;288(29):20883-95.
- Fusco G, De simone A, Gopinath T, et al. Direct observation of the three regions in α-synuclein that determine its membrane-bound behaviour. Nat Commun. 2014;5:3827.
- Bayburt TH, Sligar SG. Self-assembly of single integral membrane proteins into soluble nanoscale phospholipid bilayers. Protein Sci. 2003;12(11):2476-81.
- Bayburt TH, Sligar SG. Membrane protein assembly into Nanodiscs. FEBS Lett. 2010;584(9):1721-7.
- Li L, Chen J, Mishra VK, et al. Double belt structure of discoidal high density lipoproteins: molecular basis for size heterogeneity. J Mol Biol. 2004;343(5):1293-311.
- George JM. The synucleins. Genome Biol. 2002; 3(1): REVIEWS3002.
- Braun AR, Lacy MM, Ducas VC, Rhoades E, Sachs JN. α-Synuclein-induced membrane remodeling is driven by binding affinity, partition depth, and interleaflet order asymmetry. J Am Chem Soc. 2014;136(28):9962-72.
- Golovko MY, Barceló-coblijn G, Castagnet PI, Austin S, Combs CK, Murphy EJ. The role of alpha-synuclein in brain lipid metabolism: a downstream impact on brain inflammatory response. Mol Cell Biochem. 2009;326(1-2):55-66.
- Calo L, Wegrzynowicz M, Santivañez-perez J, Grazia spillantini M. Synaptic failure and α-synuclein. Mov Disord. 2016;31(2):169-77.
- Nakamura K. α-Synuclein and mitochondria: partners in crime?. Neurotherapeutics. 2013;10(3):391-9.
